Semaglutide vs Tirzepatide: What’s the Difference?
Learn the difference between semaglutide and tirzepatide, including how they work, key similarities, major differences, side effects, and why people often compare them.
GlobalPeptide
3/12/20265 min read


What’s the Difference Between Semaglutide and Tirzepatide?
When people start reading about newer weight-management medications, two names tend to come up again and again: semaglutide and tirzepatide. They are often discussed side by side because both are once-weekly injectable prescription medicines, and both are associated with weight reduction and improved glycemic control in the settings for which they are approved. But despite the overlap, they are not the same drug, and the difference is more than just branding.
Why Are Semaglutide and Tirzepatide Often Compared?
The comparison is easy to understand. Both drugs are widely recognized in discussions about obesity treatment and metabolic health, and both are part of a broader shift toward more effective medical therapy for chronic weight management. Semaglutide is marketed in FDA-approved products such as Wegovy and Ozempic, while tirzepatide is marketed in FDA-approved products such as Zepbound and Mounjaro. Their approved uses are not identical, but there is enough practical overlap that patients, clinicians, and readers frequently want a straightforward explanation of how they differ.
At a glance, they can seem very similar. Both are taken once weekly by subcutaneous injection. Both require dose escalation over time. Both can cause gastrointestinal side effects, especially early in treatment. And both carry boxed warnings related to thyroid C-cell tumors observed in animals.
What Is Semaglutide?
Semaglutide is a GLP-1 receptor agonist. In simple terms, it mimics the action of glucagon-like peptide-1, a hormone involved in appetite regulation, insulin secretion, and gastric emptying. FDA-approved semaglutide products include Ozempic for type 2 diabetes and Wegovy for chronic weight management in certain adults and pediatric patients, with Wegovy also carrying an indication to reduce the risk of major adverse cardiovascular events in certain adults with established cardiovascular disease and obesity or overweight.
That matters because semaglutide is not just “a weight loss shot.” Its approved labeling reflects multiple clinical roles depending on the product and patient population. This is one reason semaglutide remains such a central reference point when people compare newer therapies.
What Is Tirzepatide?
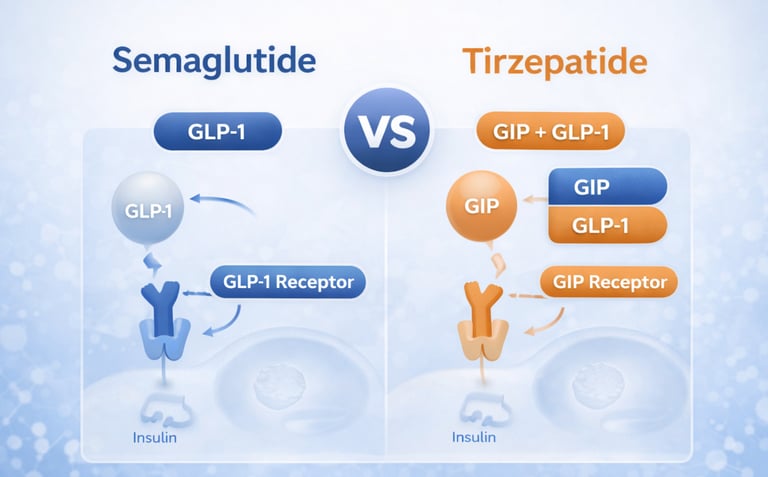
Tirzepatide works differently. It is a dual GIP and GLP-1 receptor agonist, which means it activates both glucose-dependent insulinotropic polypeptide and glucagon-like peptide-1 pathways. That dual mechanism is one of the main scientific distinctions between tirzepatide and semaglutide, and it is a major reason tirzepatide has drawn so much interest.
FDA-approved tirzepatide products include Mounjaro for type 2 diabetes and Zepbound for chronic weight management. The Zepbound label also includes obstructive sleep apnea in adults with obesity as an approved indication. Like semaglutide products, tirzepatide is dosed once weekly and titrated upward over time, starting at 2.5 mg weekly for 4 weeks before escalation.
How Do They Work in the Body?
In practical terms, both medications influence appetite, food intake, and metabolic regulation. Both are designed for long-term treatment rather than quick short-term effects. But tirzepatide’s dual-pathway activity is often cited as one reason it may achieve stronger average weight-loss outcomes in some study settings.
Key Differences Between Semaglutide and Tirzepatide
The second major difference is approved-product positioning. Wegovy and Ozempic are semaglutide products with different labeled uses, while Zepbound and Mounjaro are tirzepatide products with their own approved indications. Readers often talk about “semaglutide” and “tirzepatide” as though each were one product, but in practice the exact brand, indication, and label matter.
The third difference is the current head-to-head evidence people usually care about most: comparative outcomes. In a 2025 New England Journal of Medicine trial involving adults with obesity but without diabetes, tirzepatide was superior to semaglutide with respect to percentage change in body weight at 72 weeks. That study does not erase the value of semaglutide, but it does help explain why so many people now ask whether tirzepatide is “stronger.”
Side Effects and Tolerability
Both also carry boxed warnings about thyroid C-cell tumors observed in rodents, and both are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or in patients with Multiple Endocrine Neoplasia syndrome type 2. The relevance of the rodent thyroid findings to humans has not been established, but the boxed warning remains a prominent part of FDA labeling for both drug classes.
That is why the “better drug” conversation is never just about average weight loss. A treatment may look more potent in a trial, but tolerability, contraindications, access, and clinical context still matter in practice. This is also why a simple online comparison is useful for understanding the basics, but not enough to replace individualized medical guidance.
Which One Do People Usually Ask About for Weight Management?
In search behavior and patient conversations, these two drugs are often compared through a weight-management lens. That is understandable because Wegovy and Zepbound are both FDA-approved for chronic weight management, and because the most visible media coverage tends to focus on body-weight outcomes.
Still, it is worth keeping the framing accurate. Semaglutide has a well-established role and broad recognition, including a cardiovascular risk-reduction indication on the Wegovy label for certain adults. Tirzepatide has a newer dual-pathway mechanism and strong comparative data that suggest greater average weight reduction in at least some populations. Both are clinically important, but they are not interchangeable in every sense.
Final Thoughts
So, what is the difference between semaglutide and tirzepatide? The short answer is that semaglutide is a GLP-1 receptor agonist, while tirzepatide is a dual GIP and GLP-1 receptor agonist. They are both once-weekly injectable prescription drugs, both are widely discussed in obesity and metabolic care, and both can support meaningful clinical outcomes. But they differ in mechanism, labeling, and the comparative evidence now shaping public interest in the category.
For readers trying to understand the topic at a high level, that is the real takeaway: these drugs may belong to the same broad conversation, but they are not the same treatment. Knowing that distinction makes everything else easier to understand.
Interested in more practical articles on peptide-related topics? Explore our blog for clear comparisons, ingredient insights, and professional industry content written for a broader business audience.



Although people often focus on effectiveness, side effects are a big part of the real-world difference. Both semaglutide and tirzepatide commonly cause gastrointestinal adverse reactions such as nausea, diarrhea, vomiting, constipation, abdominal pain, and reduced appetite. These effects are one reason both drugs use gradual dose escalation rather than starting at the highest maintenance dose.


The biggest difference is still mechanism. Semaglutide is a GLP-1 receptor agonist. Tirzepatide is a dual GIP/GLP-1 receptor agonist. That single distinction explains much of why these drugs are discussed differently in clinical and commercial settings.
This is the clearest technical difference between the two drugs. Semaglutide targets one key pathway, GLP-1. Tirzepatide targets two, GIP and GLP-1. That does not mean one is automatically right and the other automatically wrong. It means they are pharmacologically distinct, even though they are often used in similar conversations.
Contact
Reach out for partnerships and inquiries
Phone
+852 4422 4956
© 2025. All rights reserved.